The HSN code for medicines in India falls under Chapter 30 of the Customs Tariff Act, with the primary heading being 3004 for medicaments consisting of mixed or unmixed products for therapeutic use in measured doses. Common medicines like paracetamol tablets are classified under 3004.90.69, while vaccines fall under 3002.20. The Basic Customs Duty on most finished medicines is 10%, with GST rates of 5% (essential medicines on NLEM) or 12% (others).
What is the HSN code for medicines in India?
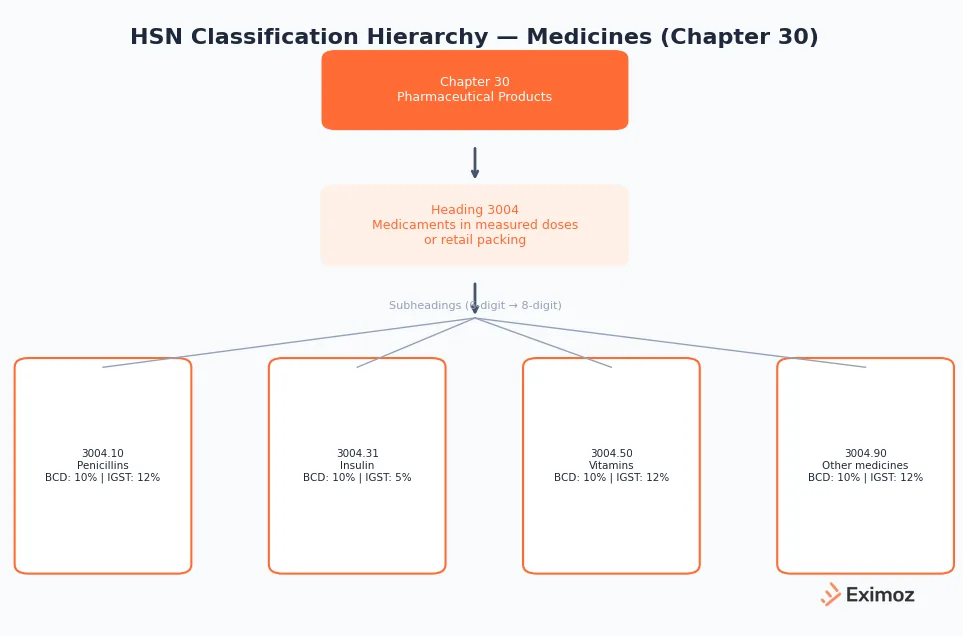
Medicines are classified under Chapter 30 of the Customs Tariff Act, titled "Pharmaceutical Products." Two headings cover most medicine imports:
- HSN 3003: Medicaments not in measured doses or retail packing (bulk formulations)
- HSN 3004: Medicaments in measured doses or retail packing
Most finished medicines at Indian ports fall under 3004, whether that is a blister strip of tablets, a pre-filled syringe, or a bottle of syrup with dosage markings.
The 8-digit code depends on the active ingredient. Paracetamol tablets are 3004.90.69. Ayurvedic medicines sit under 3004.90.11 through 3004.90.19. APIs (Active Pharmaceutical Ingredients) do not fall under Chapter 30. They are classified under Chapters 28-29 based on chemical composition.
How are different types of medicines classified under HSN?
| Medicine Type | HSN Code | BCD Rate | IGST Rate | CDSCO License Required | Key Notes |
|---|---|---|---|---|---|
| Tablets/Capsules (allopathic) | 3004.90 | 10% | 5% or 12% | Yes | 8-digit code depends on active ingredient |
| Syrups/Liquid medicines | 3004.50 / 3004.90 | 10% | 5% or 12% | Yes | Oral liquids in measured doses |
| Vaccines | 3002.20 | 10% | 5% | Yes | Human and veterinary vaccines |
| Insulin | 3004.31 / 3004.32 | 10% | 5% | Yes | Listed on NLEM, lower GST |
| Ayurvedic medicines | 3004.90.11–19 | 10% | 12% (some 5%) | No (AYUSH license) | Regulated by Ministry of AYUSH |
| Surgical dressings with medicine | 3005.10 | 10% | 12% | Case-specific | Medicated bandages, plasters |
| Veterinary medicines | 3004.50 / 3004.90 | 10% | 12% | Yes (separate category) | Animal health products |
| APIs | 2933–2942 | 2.5%–7.5% | 18% | Registration Certificate | Classified by chemical structure |
| Medical devices | Chapter 90 | Varies | 12% or 18% | Yes (MD-14/MD-15) | Separate chapter, not Chapter 30 |
| Diagnostic reagents | 3002.10 / 3822 | 10% | 12% | Case-specific | Some fall under Chapter 38 |
Source: CBIC Customs Tariff 2024-25, rates subject to annual budget notifications.
What is the customs duty on importing medicines into India?
Finished medicines under HSN 3004 attract a Basic Customs Duty (BCD) of 10%. On top of BCD, importers pay:
- Social Welfare Surcharge (SWS): 10% of BCD (effectively 1% of assessable value)
- IGST: 5% for medicines on the National List of Essential Medicines (NLEM) or 12% for others
The effective landed cost for a finished medicine import works out to roughly 16-24% above CIF value, depending on the IGST slab.
APIs attract lower BCD of 2.5% to 7.5% but carry 18% IGST. Some medicines qualify for concessional duty under specific CBIC notifications. Anti-cancer drugs, HIV/AIDS medications, and certain rare disease treatments have received full or partial BCD exemptions.
What licenses are needed to import medicines into India?
Importing medicines requires multiple approvals from CDSCO (Central Drugs Standard Control Organisation):
- Import License from CDSCO: Filed on Form MD-14 (drugs) or MD-15 (medical devices). This is the primary approval for importing a specific drug.
- Registration Certificate under Rule 27A: The overseas manufacturing facility must be registered with CDSCO.
- NOC from DCGI: For new drugs not yet approved in India, a No Objection Certificate from the Drugs Controller General is mandatory.
- Drug License from State Drug Controller: The Indian importer needs a valid wholesale drug license from their state's Drug Control department.
- IEC (Import Export Code): Standard DGFT requirement for any import activity.
For Ayurvedic, Siddha, and Unani medicines, licensing falls under the Ministry of AYUSH instead of CDSCO.
Missing any of these approvals will result in the consignment being held at port. Pharma holds are among the most expensive because temperature-controlled goods incur higher demurrage and storage charges.
What is the GST rate on medicines in India?
GST on medicines depends on whether the drug appears on the NLEM published by the Ministry of Health and Family Welfare:
- 5% GST: Medicines on NLEM, including paracetamol, metformin, amoxicillin, and insulin.
- 12% GST: Most other pharmaceutical products, including branded generics not on NLEM, Ayurvedic medicines, and veterinary drugs.
- 18% GST: APIs, intermediates, and some diagnostic reagents.
For imports, the applicable rate is IGST (Integrated GST), collected at the port. IGST paid on imports can be claimed as input tax credit.
Check the GST Rate Schedule for your specific product, since rates change with GST Council meetings. The NLEM is also updated periodically, and a drug moving on or off the list changes its GST rate.
Frequently asked questions
What is the HSN code for tablets and capsules in India?
Tablets and capsules fall under HSN 3004.90, which covers medicaments in measured doses. The 8-digit code depends on the active ingredient. Paracetamol tablets are 3004.90.69. Ayurvedic tablets and capsules have sub-headings from 3004.90.11 to 3004.90.19.
#
What is the customs duty on importing medicines into India?
BCD on most finished pharmaceutical products (HSN 3004) is 10%. Add Social Welfare Surcharge at 10% of BCD and IGST at 5% (NLEM drugs) or 12% (others). APIs under HSN 2933-2942 attract lower BCD of 2.5% to 7.5% but carry 18% IGST.
Do I need a drug license to import medicines into India?
Yes. You need an Import License from CDSCO (Form MD-14), a Drug License from your State Drug Controller, a Registration Certificate under Rule 27A for the overseas manufacturer, and for new drugs, an NOC from the DCGI.
What is the HSN code for Ayurvedic medicines?
Ayurvedic medicines are classified under HSN 3004.90.11 to 3004.90.19. GST is generally 12%, though some Ayurvedic products attract 5% GST. These are regulated by the Ministry of AYUSH, not CDSCO.
What is the difference between HSN 3003 and 3004 for medicines?
HSN 3003 covers medicines in bulk form, not in measured doses or retail packing. HSN 3004 covers medicines in measured doses or retail packing. Most imported finished medicines fall under 3004. Bulk APIs and unformulated drug mixtures may fall under 3003 or Chapters 28-29.
Pharma imports have the strictest compliance requirements of any product category in Indian customs. Between HSN classification, CDSCO licensing, state drug controller approvals, and GST rate verification, there are more places for things to go wrong than in almost any other import category. Eximoz maps your products to the right HSN codes and flags CDSCO and drug license requirements before you ship.


